Client
Rangel
Sector
Services
Techstack
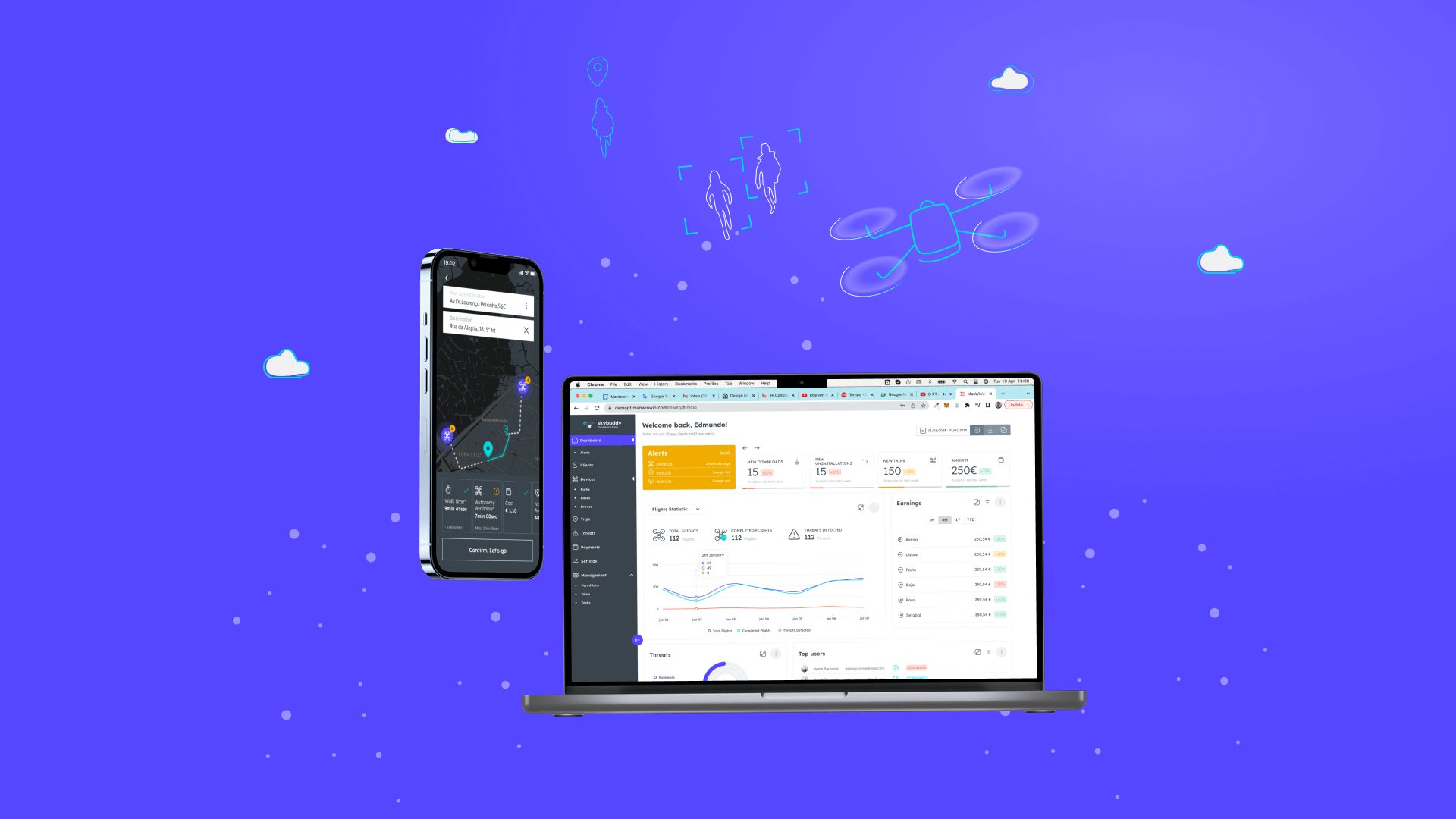
Rangel - MeDS
A Medicine distributed solution for healthcare professionals and patients.
Challenge
Rangel, an international pharmaceutical logistics company headquartered in Portugal, specializes in the transportation and storage of pharmaceuticals in more than 220 countries and territories. Recognizing the need to keep pace with rapidly evolving industry trends and advancements, Rangel wanted to integrate digital innovation and transformation into its operations. To achieve this goal, the company turned to LOAD for assistance in developing an innovative MedTech solution. The solution was to be developed with a human-centric approach focused on the accuracy and reliability of medical information.
Research & Development
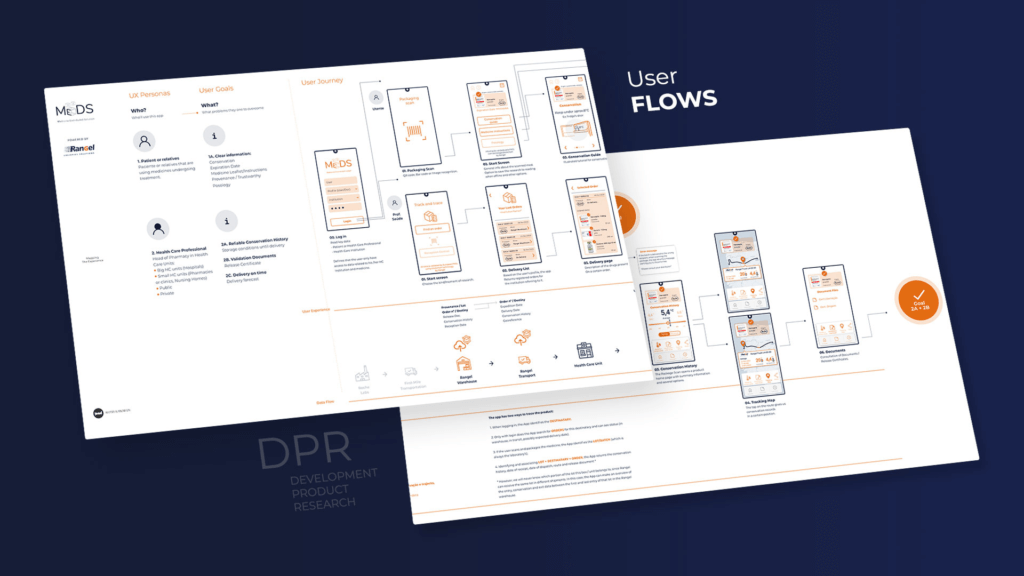
To determine the optimal solution for pharmaceutical logistics requirements, we initiated a comprehensive Digital Product Research (DPR) process. Recognizing the challenges of addressing complex issues and unknown factors, we began by collecting all relevant assumptions related to the product or service. Through this reflection exercise, we developed a methodology based on design thinking and technical research. Digital Product Research is a highly structured methodology consisting of fourteen different phases. In a co-creation process, we work with our clients to identify all relevant information about their existing product and their future goals.
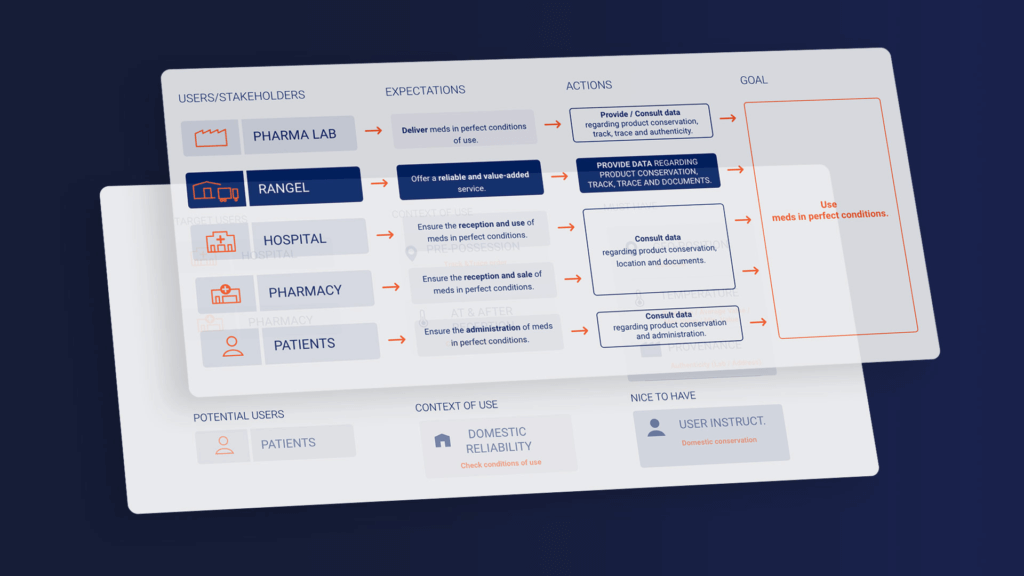
We began by conducting a stakeholder analysis that gave us a comprehensive understanding of the client’s vision and overall business. This included one-on-one meetings with each stakeholder to identify their specific needs and challenges. We then collated this information to brainstorm with the client, and started developing a user journey map. The user journey map serves as a concise representation of the user’s interaction with the product or service and provides a step-by-step breakdown from the user’s perspective, while also detailing any potential obstacles that may arise.
To deepen our understanding of the relevant technologies and potential implementations, we conducted a thorough benchmarking, technical research, and risk assessment. In the process, we identified two key findings and recommendations. Firstly, we strongly recommended the use of decentralization due to the high-end nature of the medicines involved. Secondly, we recommended incorporating blockchain technology, which offers a transparent and genuine approach to information dissemination. By leveraging the inherent architecture of blockchain, we can ensure the quality of the services and information provided to users.
Solution
MeDS is a blockchain-based solution that embodies a Human Centricity approach. This approach aims to provide patients and healthcare professionals with the most up-to-date information about their medications, regardless of their location.
This approach allows to validate drug traceability by sharing important information such as source (labs), destination, temperature, humidity, and timestamp. The information will be available to pharmacists, physicians and patients, ensuring they have access to all relevant data. By using blockchain technology, MeDS can provide a decentralized and transparent platform that ensures all data exchanged is genuine, accurate and up-to-date. Ultimately, this approach allows us to deliver a secure and reliable solution that prioritizes the needs of patients and healthcare professionals alike.

Explore More
Related Projects